|
Abstract
NAT catalyzes the addition of an acetyl group from acetyl-CoA to terminal nitrogen on substrates such as isoniazid and sulfamethazine and these were termed “polymorphic” substrates for N-acetylation. The present project was designed to determine the NAT2 acetylation in local human population by using probe drugs like sulfmathazine, caffeine, isoniazid and Dapson under indigenous conditions. Blood and urine samples of the human volunteers were collected at different time intervals after drug administration. The acetylation status was determined both by phenotypically (HPLC assay) and genotypically by PCR based allel specific amplification assay. Correlation between two was determined. Biochemical parameters like complete blood picture (CBP), ALT, AST, lipid profile, Total protein, creatnine and urea level of the volunteers will be recorded. This would also help for clinical investigations under indigenous conditions. This study was used to determine the acetylation status of NAT2 under our environmental conditions, nutritional status and genetic make-up and it was used to determine dose regimen.
Problem statement
Molecular heterogeneity or genetic polymorphism of enzymes is the change in alleles of the genes for the enzyme, which results in the production of enzyme protein with variable enzyme activity and stability causing sub-therapeutic responses or no response to normal doses. Genetic polymorphism of drug metabolizing enzymes may classify a population into subgroups as slow and fast metabolizers which differ in their ability to biotransform certain drugs as caffeine, dapsone, sulphonamides and isoniazid (INH). Phenotyping and genotyping studies should be conducted prior the drug therapy for the safety of the humans.
Objectives
- The use of metabolic phenotyping or genotyping will provide more precise information for determining effective therapeutic dose for drugs undergoing acetylation.
- NAT2 is important in determinant of both the clinical efficacy and the toxicity of drugs and environmental chemicals.
- The routine genotyping before drug therapy may enable the identification of responders, nonresponders or patients at increased risk of toxicity.
Methodology
The study is being conducted on healthy human male and female volunteers of age greater than 18 years and demographic data that include the body weight, age, body temperature and blood pressure of each volunteer is also recorded. The biochemical parameters include complete blood picture, lipid profile (By kit method), lactate dehydrogenase (LDH), alanine aminotransferase (ALT), aspartate transferase (AST), total protein, albumin globulin creatnine and urea level of the volunteers is determined by the given kit methods. Phenotype was determined in a cross over design i.e at first one drug at a prescribed dose is given to the volunteers and their blood and urine samples were taken in a predetermined time intervals from 0-6 h. Reproducibility of the method is also determined in a selected number of volunteers. The free drug and acetylated metabolites of these drugs were determined by HPLC method. The phenotype is determined by measuring the molar ratio of two unchanged drug and acetylated metabolites. The genotype was determined by taking the blood samples of the volunteers and DNA was separated by standard protienase K method protocol). The wild type allele and variant alleles for NAT2 (N-acetyletransferase2) was determined by PCR based amplification of the alleles in DNA by using the specific primers for the respective mutant alleles. The correlation was determined between the phenotype and genotype is determined by appropriate statistical method. (Steel et al., 1997).
 |
|
Results and Discussion
Analytical methods for probe drugs and their acetyl metabolites by HPLC were developed and validated. These drugs include sulfamethazine, isoniazid, dapsone and caffeine. Standards and replicate QC samples of plasma and/ or urine at each concentration were analyzed on three consecutive days, after which inter- and intra-day means, standard deviations (S.D.), and coefficients of variation (CV%) were calculated by standard methods ( ICH Guidelines, 2005). At the LOQ in plasma and urine, the signal to noise ratio was greater than 5:1. The standard curves for all probe drugs and their acetyl metabolites were constructed in the range of 5-100 µg/ml and found to be linear with a slope ranging between 0.006 to 0.065, y-intercept of − 0.017 to + 0.178 and a correlation coefficient of 0.978 to 0.999. The intra- and inter-day precision and %CV for all probe drugs and their acetyl metabolites were ≤ 10.0 % at low quality control (LOQ). The biochemical parameters were determined in 50 volunteers. The average ± SD (mg/dL) for serum urea and creatnine were 28.2 ± 5.92, and 0.786 ± 0.188 in female whereas 36.9 ± 8.14 and 1.05 ± 0.317 in males respectively. The average ± SD (mg/dL) for total cholesterol, triglycerides, HDL and LDL were 174 ± 46.1, 119 ± 42.4, 60.1 ± 12.5 and 58.7 ± 38.3 in female whereas 207.9 ± 59.2, 141 ± 42.2, 85.1 ± 25.5 and 56.1 ± 38.9 in males respectively. The average ± SD (mg/dL) of haemoglobulin, total protein, albumin, globulin were 6.85 ± .305, 5.32 ± 0.314 and 1.53 ± 0.271 in female and 7.18 ± 0.299, 5.96 ± 0.196 & 1.22 ± 0.240 in males. The enzyme levels (ALT & AST) were 25.7 ± 6.84, 34.1 ± 7.88 in females and 28.8 ± 7.4 and 40.6 ± 6.14 U/L in males. The plasma samples were used for HPLC analysis to determine the acetylator phenotype of the volunteers after oral administration of 500 mg of sulfamethazine. It was found that slow acetylators were 80 and 72 % whereas fast acetylators 20 and 28 % in female and male volunteers, respectively. Our population was found to be slow acetylator for sulphamethazine.
Conclusions
It was found that slow acetylators were 80 and 72 % whereas fast acetylators 20 and 28 % in female and male volunteers, respectively. Our population was found to be slow acetylator for sulphamethazine.
Recommendations
The use of metabolic phenotyping or genotyping will provide more precise information for determining effective therapeutic dose for drugs undergoing acetylation.
One to two Photographs of Project
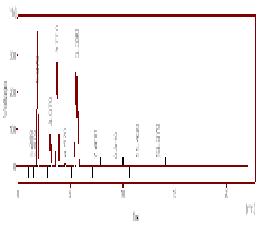
Chromatograms for High Quality Control (HOQ) 80 µg/mL in Plasma containing Internal standard
( Sulfadiazine, SDZ), Sulfamethazine (SMZ) and Acetyl Sulfamethazine ( AcSMZ) in plasma The retention time (minutes) for sulfadiazine ( 3.62), sulfamethazine ( 8.57) and acetylsulfamethazine ( 10.27)
|